
TITLE
Immunomodulatory inhibition of osteoclastogenesis by a marine microalgal ethanol fraction targeting T-cells, antigen presentation, and macrophage fate
JOURNAL
Frontiers in Immunology
AUTHORS
Alessio Carletti, Katia Pes, , Joana T. Rosa, , Hugo Galvão Pereira, Bruno Louro, M. Leonor Cancela, Vincent Laizé, Paulo J. Gavaia
ABSTRACT
Background: Targeting immune pathways to prevent bone loss represents a promising, yet underexplored therapeutic strategy.
Methods: An ethanol-soluble fraction derived from the freeze-dried biomass of the marine microalga Skeletonema costatum (SKLT) was tested for its ability to modulate immune responses and inhibit osteoclastogenesis. Its effects were evaluated in a zebrafish model of bone regeneration, a medaka model of RANKL-induced osteoporosis, and in vitro using murine RAW 264.7 macrophages. Transcriptomic profiling of regenerating fin blastemas at 24 hours post-amputation was performed to identify the affected molecular pathways.
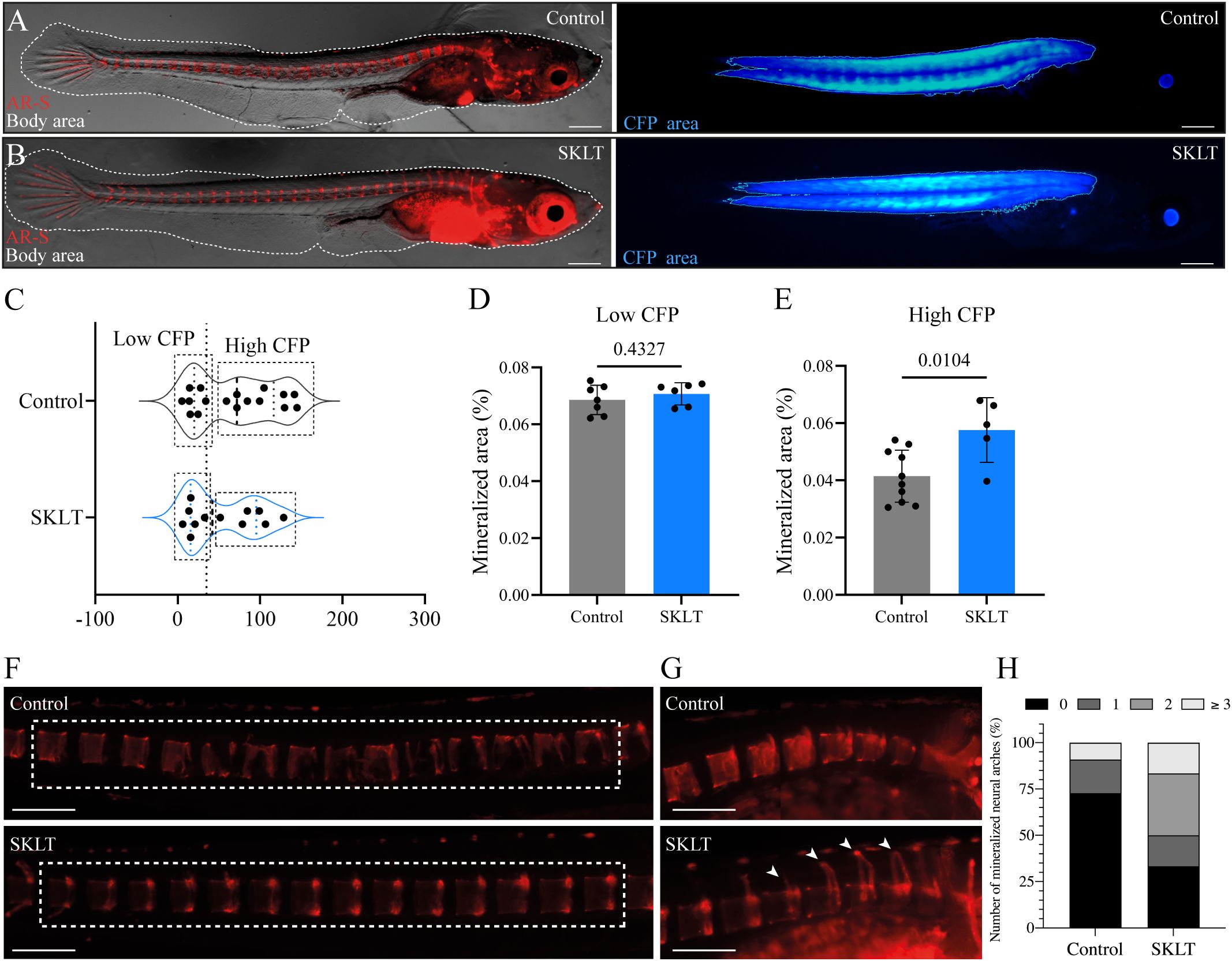
Results: In zebrafish, SKLT treatment suppressed the recruitment of osteoclast precursors and altered mineralization dynamics. Transcriptomic profiling revealed downregulation of genes involved in inflammation, antigen presentation, T-cell activation, and macrophage commitment towards osteoclastogenesis, accompanied by reduced expression of chemokines and cytokines that promote osteoclast precursor recruitment and fusion. In medaka, SKLT significantly reduced vertebral bone loss and enhanced neural arch mineralization in larvae with high RANKL expression. In vitro, SKLT inhibited proliferation and osteoclastic differentiation of murine RAW 264.7 macrophages exposed to RANKL without inducing cytotoxicity.
Conclusion: These findings identify S. costatum as a source of bioactive immunomodulatory compounds capable of interfering with key osteoimmune mechanisms. Beyond providing proof of concept for their therapeutic potential in bone erosive disorders, this work opens avenues for isolating and characterizing the active molecules, optimizing their delivery, and evaluating their efficacy in preclinical mammalian models. Such strategies could expand the repertoire of safe, nutraceutical-based or adjuvant therapies for osteoporosis and other inflammation-driven skeletal diseases, complementing and potentially enhancing current antiresorptive and anabolic treatments.



